The Fentanyl Vaccine Is Here: Inside the Shot That Could End America's Deadliest Epidemic

The vaccine approach to addiction treatment represents a fundamental shift in how medicine fights the opioid crisis. Photo: Pexels
Somewhere in a clinical research facility in Leiden, the Netherlands, roughly 40 healthy adults are rolling up their sleeves for a shot that nobody imagined would exist a decade ago. They are not being vaccinated against a virus. They are being vaccinated against a drug - specifically, against the synthetic opioid that has killed more Americans than any other substance in history.
The experimental vaccine, developed by researchers at the University of Houston and licensed by biotech startup ARMR Sciences, trains the human immune system to produce antibodies that intercept fentanyl molecules in the bloodstream before they ever reach the brain. No euphoria. No respiratory depression. No overdose. If it works in humans the way it worked in rats, it would become the first proactive treatment for fentanyl addiction - a shot that prevents the crisis rather than responding to it after the damage is done.
This is not theoretical. Phase I/II human trials began in early 2026 at the Centre for Human Drug Research in the Netherlands. Bloomberg reported this week that the trial is now underway, with initial safety and dosing data expected within months. The science is peer-reviewed. The funding came from the U.S. Department of Defense. And the timing could not be more urgent.
Over 600,000 Americans have died from opioid overdoses since 1999, according to the Centers for Disease Control and Prevention. Fentanyl alone now accounts for roughly 69% of all overdose deaths. The drug is 50 times more potent than heroin. A lethal dose weighs about two milligrams - the equivalent of 12 grains of salt.
The question is no longer whether the science can produce a fentanyl vaccine. The question is whether anything can move fast enough to matter.

The scale of destruction: 600,000+ deaths, $1.5 trillion in economic costs, and a settlement that barely scratched the surface. BLACKWIRE infographic
The Epidemic That Won't Die

Fentanyl has saturated America's drug supply, appearing in everything from counterfeit pills to cocaine and methamphetamine. Photo: Pexels
The American opioid crisis did not begin with fentanyl. It began with a lie. In 1996, Purdue Pharma launched OxyContin with a marketing campaign that told doctors their new extended-release opioid carried a low risk of addiction. Internal documents later revealed the company knew this was false. By the time Purdue pleaded guilty to federal criminal charges in 2007 - paying a $600 million fine - millions of Americans were already hooked.
The first wave of the epidemic was prescription opioids: OxyContin, Vicodin, Percocet. Doctors wrote more than 250 million opioid prescriptions per year at the peak, enough for every adult in America to have a bottle. When regulators finally cracked down on pill mills and prescription pads in the early 2010s, addicted patients did not simply get better. They turned to heroin, which was cheaper and easier to find. That was wave two.
Wave three arrived around 2013, when illicitly manufactured fentanyl began flooding the market from Chinese chemical suppliers and Mexican cartels. Fentanyl's economics are devastating in their simplicity: it is cheap to synthesize, requires tiny quantities per dose, and can be mixed into virtually any drug supply. By 2022, overdose deaths in the United States hit a staggering 109,680 - a number so large it exceeds the American death toll of the entire Vietnam War.
The crisis has since eased somewhat. Provisional CDC data from March 2026 shows an estimated 71,542 overdose deaths in the 12 months ending October 2025, a 17.1% decline from the prior year. But even that "improved" figure means roughly 196 Americans are dying every single day from drug overdoses. The crisis has not ended. The body count has merely become less apocalyptic.
Fentanyl's penetration into the drug supply has made the crisis qualitatively different from anything that came before. It is no longer just an opioid problem. Fentanyl now contaminates cocaine, methamphetamine, counterfeit prescription pills, and even marijuana in some markets. People who have never intentionally used an opioid are dying because a single contaminated pill contains a lethal dose of fentanyl. Teenagers are dying because pills purchased on social media turn out to be pressed fentanyl tablets.
The existing medical toolkit, while valuable, has clear limitations. Naloxone (brand name Narcan) can reverse an overdose, but only if someone is present to administer it in time. Medications for opioid use disorder - buprenorphine (Suboxone), methadone, and naltrexone - are effective but require ongoing compliance, regular clinic visits, and a patient who wants to stop using. None of them prevents the first overdose. None of them works if the user does not know they have consumed fentanyl.
For decades, addiction medicine has been reactive. A vaccine that works proactively - that protects the body before fentanyl ever reaches the brain - would represent the most significant paradigm shift in addiction treatment since the invention of methadone in the 1960s.
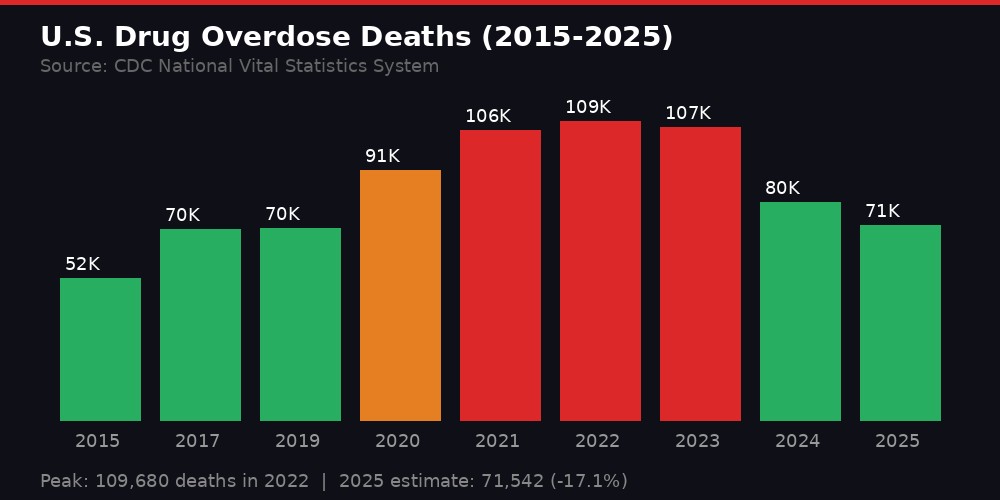
The rise and partial retreat: overdose deaths peaked at nearly 110,000 in 2022 before declining. BLACKWIRE infographic
How You Vaccinate Against a Drug

The challenge: fentanyl molecules are too small for the immune system to detect on their own. Researchers had to engineer a workaround. Photo: Pexels
The concept sounds counterintuitive. Vaccines fight pathogens - viruses, bacteria, parasites. How do you vaccinate against a molecule?
The answer lies in a technique called hapten conjugation. Fentanyl is a tiny molecule, far too small for the immune system to recognize on its own. Unlike a virus, which is large and studded with proteins that immune cells can latch onto, fentanyl slips through unnoticed. The immune system simply does not register it as a threat.
Dr. Colin Haile, a pharmacologist at the University of Houston and co-founder of ARMR Sciences, solved this problem by chemically linking a synthetic fragment of fentanyl to a much larger carrier protein called CRM197. CRM197 is a deactivated form of diphtheria toxin already used safely in several existing vaccines, including those for pneumococcal disease. When the immune system encounters the CRM197-fentanyl conjugate, it mounts a robust response against the entire complex - including the fentanyl component attached to it.
"Our goal as a company is to eliminate the lethality of the drug supply. We want to go about doing that by attacking the root cause of not only addiction, but also, obviously, overdose."- Colin Gage, co-founder and CEO, ARMR Sciences (via Fox News, January 2026)
The vaccine also includes an adjuvant called dmLT, a modified E. coli toxin that further amplifies the immune response. This three-component cocktail - fentanyl fragment, carrier protein, and adjuvant - teaches the body to produce specialized anti-fentanyl antibodies that persist in the bloodstream for months.
When a vaccinated person encounters real fentanyl, these antibodies bind to the drug molecules before they can cross the blood-brain barrier. Fentanyl locked to an antibody is too large to pass through. Instead of reaching opioid receptors in the brain and triggering euphoria and respiratory depression, the fentanyl is routed to the kidneys and eliminated from the body.
The result, at least in animal studies, is striking. Vaccinated rats showed no signs of fentanyl intoxication even when given doses that would normally be fatal. The antibodies did not interfere with other opioid medications, meaning a vaccinated patient could still receive morphine, oxycodone, methadone, or buprenorphine for pain management or addiction treatment.
"It's similar to how we design vaccines for COVID-19 or influenza. We want the body to recognize the drug as a threat and mount a defense."- Dr. Chenming "Mike" Zhang, Virginia Tech, biological systems engineering professor (via WVTF, March 2026)
Separately, researchers at Virginia Tech led by Professor Chenming "Mike" Zhang have published complementary work in the Journal of Controlled Release showing that a novel hybrid lipid-polymer nanoparticle vaccine produced even stronger immune responses in mice. Zhang's approach coats biodegradable nanoparticles - designed to mimic the size and structure of viruses - with opioid molecules, creating a delivery vehicle that the immune system attacks aggressively. Early results suggest antibody levels could remain effective for several months, with optimization potentially extending protection to a full year.
The two research teams represent different engineering approaches to the same core idea: train the immune system to neutralize fentanyl before it kills.

The four-step mechanism: vaccination, immune response, fentanyl interception, brain protection. BLACKWIRE infographic
The Human Trial: 40 Volunteers in Leiden

Phase I trials are underway at the Centre for Human Drug Research in the Netherlands. Photo: Pexels
The Phase I/II trial launched in early 2026 at the Centre for Human Drug Research (CHDR) in Leiden, the Netherlands. CHDR is one of Europe's leading clinical pharmacology research institutes, specializing in first-in-human studies. The decision to run the trial in the Netherlands rather than the United States was partly practical - CHDR has extensive experience with drug challenge studies, where participants receive controlled doses of substances to test whether a treatment works.
The trial will enroll approximately 40 healthy adult volunteers and proceed in two stages. In the first stage, participants receive a series of two vaccine shots in varying doses. Researchers will draw blood at regular intervals to measure anti-fentanyl antibody levels and monitor for adverse effects. This is the standard safety assessment that every new drug must pass before moving forward.
If the safety data is clean, the trial moves to its second and more dramatic stage. Some participants will be given medical-grade fentanyl under close clinical supervision. Researchers will then measure whether the vaccine successfully blocks fentanyl's effects - specifically, whether it prevents the respiratory depression that kills overdose victims and the euphoric high that drives addiction.
This is where the trial design gets bold. Giving fentanyl to human subjects, even under medical supervision, carries inherent risk. But it is also the only way to definitively prove the vaccine works. Animal studies can only tell you so much. The FDA, and the scientific community at large, will demand proof that antibodies generated in human subjects can actually intercept fentanyl at clinically meaningful concentrations.
ARMR Sciences has not publicly disclosed a timeline for reporting initial results, but Phase I trials typically run 6 to 12 months. If safety and immunogenicity data are favorable, Phase II trials testing efficacy in larger populations would follow. Phase III trials, the final step before FDA approval, would require hundreds or thousands of participants and could take several years.
The realistic timeline for a commercially available fentanyl vaccine, assuming everything goes well, is likely 2030 or beyond. That is an eternity in a crisis that kills 196 people per day. But the alternative - no vaccine at all - is worse.
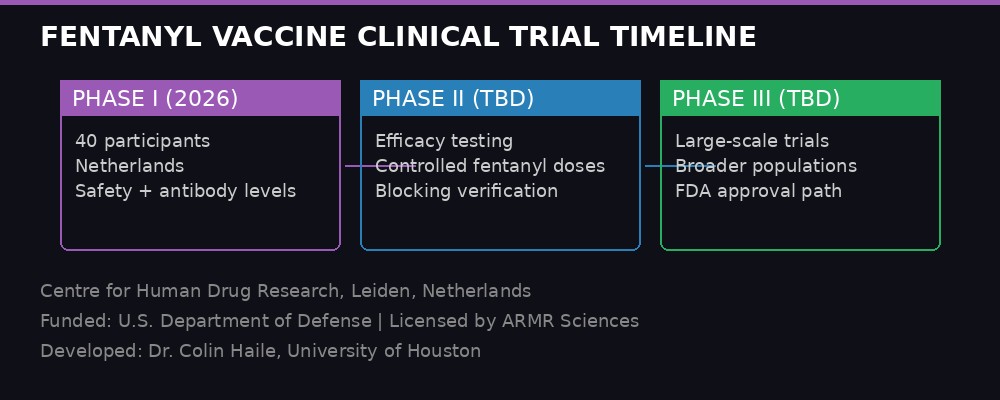
The road ahead: Phase I is underway, but FDA approval could still be years away. BLACKWIRE infographic
Who Gets the Shot - and Who Decides

The vaccine is not designed for general population use. Its target: people with opioid use disorder and those at high risk of accidental exposure. Photo: Pexels
If the vaccine eventually reaches the market, the question of who receives it will be as complex as the science itself. Unlike a flu shot or a COVID vaccine, the fentanyl vaccine is not intended for universal deployment. It is a targeted intervention for specific populations.
The most obvious candidates are people with diagnosed opioid use disorder - roughly 2.7 million Americans, according to the Substance Abuse and Mental Health Services Administration (SAMHSA). For patients struggling with relapse, the vaccine could provide a biological safety net. Even if they use fentanyl, the antibodies would block the high and prevent overdose, buying time for treatment and recovery.
A second population is at-risk youth. Dr. Zhang at Virginia Tech has been particularly vocal about this application. Adolescents and young adults are disproportionately likely to try drugs for the first time, often without understanding the risks. A teenager who takes a counterfeit Percocet at a party does not know it contains fentanyl. If vaccinated, they would not experience the euphoric high that triggers addiction - and they would survive the encounter.
"Some youth may be vulnerable to trying opioids. If they're vaccinated, they won't experience the high, which could deter future use."- Dr. Chenming "Mike" Zhang, Virginia Tech (via Virginia Tech News, March 2026)
A third group is first responders. EMTs, police officers, and firefighters who encounter fentanyl in the field face occupational exposure risks. While the actual danger of skin-contact fentanyl exposure has been debated among toxicologists, the vaccine could provide an additional layer of protection for personnel handling seized drugs or treating overdose patients.
But the ethical terrain is treacherous. Could employers mandate the vaccine for workers in high-risk environments? Could courts order vaccination as a condition of parole or probation? Could parents vaccinate children without their consent? These questions do not have easy answers, and they will collide with a post-COVID political landscape in which vaccine skepticism has become deeply entrenched across the ideological spectrum.
The Department of Defense's involvement in funding the original research adds another layer of complexity. The military has its own opioid problem - rates of prescription opioid misuse among active-duty service members and veterans exceed civilian rates. A vaccine that protects deployed troops from weaponized fentanyl exposure or reduces addiction rates among veterans could have national security implications.
There is also the question of insurance coverage. A vaccine requires manufacturing, distribution, cold chain storage, and administration by medical professionals. If it costs hundreds of dollars per dose, will Medicaid cover it? Will private insurers? Will the populations most in need - people experiencing homelessness, people without health insurance, people in rural areas with no addiction treatment infrastructure - actually be able to access it?
History offers cautionary lessons. Naloxone, the overdose reversal drug, has been available since the 1970s. It took decades to make it widely accessible, and even now, distribution remains patchy in many parts of the country. Buprenorphine, the gold-standard medication for opioid addiction, was approved in 2002 but remains underutilized because of bureaucratic prescribing restrictions that were only fully lifted in 2023. The science is necessary. It is not sufficient.
The Skeptics and the Obstacles

Previous attempts at addiction vaccines have failed in clinical trials. The ARMR approach addresses several known failure points. Photo: Pexels
Fentanyl is not the first drug researchers have tried to vaccinate against. The concept dates back to the 1970s, when scientists first proposed antibody-based approaches to blocking drugs of abuse. Over the intervening decades, vaccines targeting nicotine, cocaine, methamphetamine, and heroin have all been developed and tested in animals. Several reached human trials.
None made it to market.
NicVAX, a nicotine vaccine developed by Nabi Biopharmaceuticals, reached Phase III trials in 2011 before failing to demonstrate efficacy. A cocaine vaccine tested at Baylor College of Medicine in 2009 produced antibodies in only 38% of participants - not enough to reliably block the drug's effects. An oxycodone vaccine developed at the Scripps Research Institute showed promise in animals but has not yet advanced to human trials.
The consistent problem has been generating a strong enough and sustained enough antibody response. Small molecule drugs like nicotine and cocaine are poor immunogens - they simply do not trigger the kind of aggressive immune response that a virus does. Previous vaccines used older conjugation techniques and adjuvants that produced antibodies, but not enough of them, and not for long enough.
ARMR Sciences claims its vaccine addresses these historical shortcomings. The use of CRM197 as a carrier protein, combined with the dmLT adjuvant, is designed to produce higher antibody titers that persist longer. In rat studies, the vaccine generated robust anti-fentanyl antibody levels that remained effective for at least six months after the initial vaccination series. Human immune responses do not always mirror animal results, but the researchers are cautiously optimistic.
Virginia Tech's nanoparticle approach takes a different tack. By engineering particles that physically resemble viruses in size and surface structure, Zhang's team is essentially tricking the immune system into responding to drug molecules as if they were a viral threat. This biomimicry strategy has shown stronger immune activation in mice compared to older conjugation methods.
Another concern is the phenomenon of "competitive dosing." A determined user could theoretically overwhelm the vaccine's antibody protection by consuming very large doses of fentanyl. If enough fentanyl molecules flood the bloodstream simultaneously, they could saturate all available antibodies, with the excess crossing into the brain. This means the vaccine may not be foolproof against intentional high-dose use - though it would still protect against accidental exposure and moderate doses.
The funding environment presents its own challenges. The current federal administration has shown limited appetite for new vaccine research funding, and the broader political climate around vaccines has become polarized since the COVID-19 pandemic. Whether Congress would allocate resources for fast-tracking a fentanyl vaccine - even one that could save tens of thousands of lives annually - is an open question.
Still, the private sector is moving. ARMR Sciences secured its initial funding and is running its trial without federal support. If Phase I data is strong, investment capital is likely to follow. The opioid treatment market generates over $3 billion annually in the United States alone. A working vaccine would be, in addition to a medical breakthrough, a commercial blockbuster.
Narcan vs. Vaccine: Reactive vs. Proactive

Naloxone reverses overdoses after they happen. The vaccine would prevent them from happening in the first place. Photo: Pexels
The comparison between naloxone and the fentanyl vaccine illuminates the fundamental shift in strategy. Naloxone is a competitive opioid receptor antagonist - it works by binding to opioid receptors in the brain and displacing fentanyl. But naloxone must be administered after the overdose has already begun, typically by a bystander who recognizes the symptoms: blue lips, shallow breathing, unresponsiveness. It works fast - within minutes - but its window of effectiveness is narrow. If nobody is around to administer it, naloxone is useless.
The vaccine operates on a completely different principle. Instead of competing with fentanyl at the brain's opioid receptors, it intercepts fentanyl in the bloodstream - upstream of the brain entirely. The antibodies act as molecular sentries, grabbing fentanyl molecules before they can cross the blood-brain barrier. No fentanyl in the brain means no euphoria, no respiratory depression, no overdose.
This is not an either-or proposition. Naloxone remains indispensable as an emergency intervention. The vaccine, if approved, would serve a different role - long-term prophylactic protection for people at elevated risk. The two approaches are complementary, not competitive.
But the symbolic difference matters. Naloxone represents a crisis paradigm: people are dying, and we are scrambling to save them one at a time. The vaccine represents a prevention paradigm: we can stop the crisis before it reaches individual bodies. After two decades of reactive interventions - pill lockboxes, prescription drug monitoring programs, drug take-back days, naloxone distribution, fentanyl test strips - the possibility of genuine prevention is, for many in addiction medicine, almost disorienting in its ambition.
The concept also extends beyond opioids. If the fentanyl vaccine succeeds, it validates the broader platform of anti-drug vaccines. The same immunological principles could be applied to methamphetamine, cocaine, or novel synthetic drugs that have not yet emerged. ARMR Sciences has publicly stated its intention to develop a multi-drug vaccine platform if the fentanyl candidate succeeds.
Dr. Yuanzhi Bian, another Virginia Tech researcher involved in the nanoparticle vaccine work, has emphasized that the vaccine is not a standalone cure. It does not address the social, economic, and psychological drivers of addiction. It does not replace therapy, housing, job training, or community support. What it does, potentially, is keep people alive long enough to access those resources.
"I think our vaccine can be a complement to the existing treatment methods. It's a completely different approach. We target the molecules in the circulation before they enter the brain. I think it's a promising approach."- Dr. Yuanzhi Bian, Virginia Tech (via WVTF, March 2026)
The Road Forward

The ultimate challenge is not science - it is delivery. Getting a working vaccine to the people who need it most will require systemic change. Photo: Pexels
The next 12 to 18 months will determine whether the fentanyl vaccine moves from promising science to viable medicine. If ARMR Sciences reports favorable safety data from its Phase I trial - no serious adverse events, measurable anti-fentanyl antibody production - the momentum will accelerate. If Virginia Tech's nanoparticle approach shows superior immune activation in subsequent animal studies, it could enter its own human trial pipeline.
The FDA has signaled willingness to fast-track treatments addressing the opioid crisis. Naloxone received over-the-counter approval in 2023 via an expedited review process. A fentanyl vaccine could potentially receive breakthrough therapy designation, which would shorten the regulatory timeline by allowing rolling submission of clinical data and more frequent FDA consultations.
But fast-tracking still takes time. Even with accelerated review, the most optimistic projections suggest a commercially available vaccine is four to six years away. In that span, at current death rates, another 250,000 or more Americans will die from drug overdoses. The vaccine will not save them.
What it can do, if the science holds, is fundamentally alter the trajectory of the crisis for the generation that follows. A world in which at-risk individuals can receive a shot that protects them from fentanyl overdose for months at a time is a world with dramatically fewer funerals, fewer orphaned children, fewer shattered families. It is a world in which addiction treatment starts from a position of safety rather than desperation.
The Sackler family made $13 billion from OxyContin before agreeing to pay $7.4 billion in settlements for their role in fueling the epidemic. The pharmaceutical industry created this crisis through greed and deception. Whether a different corner of that industry - a Houston pharmacologist, a Netherlands clinic, a startup with 40 volunteers - can produce the antidote is the most consequential medical question in America right now.
Forty people in Leiden have rolled up their sleeves. The rest of us wait.
Get BLACKWIRE reports first.
Breaking news, investigations, and analysis - straight to your phone.
Join @blackwirenews on Telegram