The Pill That Kills the Needle: FDA Greenlights Eli Lilly's Foundayo and the GLP-1 Revolution Goes Oral
A once-daily tablet approved in just 50 days. No food restrictions. No injections. Starting at $149 a month. The obesity drug war just went nuclear.

The era of the weight-loss pill has arrived. Photo: Pexels
On April 1, 2026, the U.S. Food and Drug Administration approved Foundayo, Eli Lilly's once-daily GLP-1 pill for chronic weight management in adults with obesity or overweight with weight-related medical conditions. The decision, delivered through a brand-new expedited review pilot program in just 50 days, represents the fastest obesity drug approval in FDA history and sets the stage for what analysts are calling the most consequential shift in pharmaceutical history since the statin revolution of the 1990s.
The drug's chemical name is orforglipron. Its brand name is Foundayo, pronounced "fown-DAY-oh." And its implications stretch far beyond the medicine cabinet. This is a small-molecule, non-peptide oral GLP-1 receptor agonist that can be taken at any time of day, with or without food, with or without water. No empty-stomach requirements. No morning-only dosing windows. No refrigeration. No needles.
It ships from LillyDirect starting April 6. Prescriptions are already being accepted. Retail pharmacies and telehealth platforms follow shortly after. Starting price: $149 per month for self-pay patients on the lowest dose. Commercial insurance holders with a Lilly savings card can pay as little as $25 per month. Medicare Part D enrollees will access it for $50 per month beginning July 1, 2026, under the TrumpRx platform deal brokered last November.
This is not incremental progress. This is the moment the needle era starts dying.
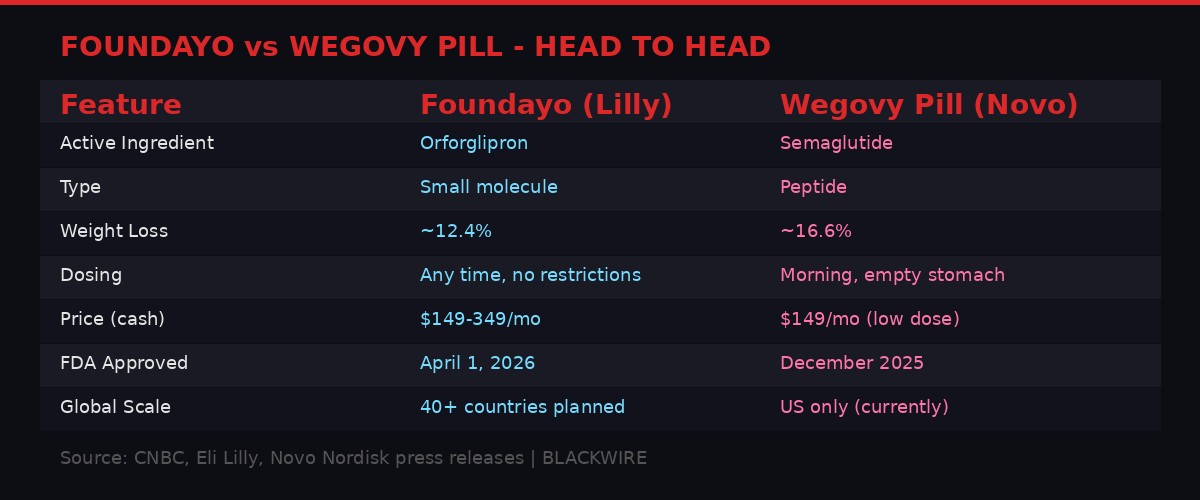
Head-to-head: Foundayo versus Novo Nordisk's Wegovy pill. Graphic: BLACKWIRE
The Science: How Foundayo Works and Why It's Different

Foundayo represents a fundamentally new molecular approach to GLP-1 therapy. Photo: Pexels
GLP-1 receptor agonists mimic a naturally occurring gut hormone called glucagon-like peptide-1. When you eat, your intestines release GLP-1, which signals the pancreas to produce insulin, slows gastric emptying, and tells your brain you're full. The injectable GLP-1 drugs that have dominated headlines since 2021 - Ozempic, Wegovy, Mounjaro, Zepbound - are all peptide-based. They're chains of amino acids engineered to last longer in the body than natural GLP-1, which breaks down within minutes.
Foundayo is architecturally different. It is a small molecule, not a peptide. This distinction matters enormously for manufacturing, distribution, and patient experience. Peptide drugs require complex biologic manufacturing processes, cold-chain storage, and specialized injection delivery systems. Small molecules can be manufactured using conventional pharmaceutical chemistry, pressed into tablets, and stored at room temperature. The production economics are fundamentally different.
Eli Lilly licensed the orforglipron molecule from Japanese pharmaceutical company Chugai in 2018, paying a reported $50 million upfront for global rights. At the time, oral GLP-1 therapy was considered a moonshot. Novo Nordisk had been working on oral semaglutide for years, and the early versions required elaborate dosing restrictions - patients had to take the pill first thing in the morning, on a completely empty stomach, with no more than four ounces of water, and then wait at least 30 minutes before eating or drinking anything else, or taking any other medication.
Orforglipron sidesteps all of that. Because it is a non-peptide small molecule, it survives the gastrointestinal tract without the protective formulation requirements that peptide-based oral drugs need. Patients can take it at breakfast, at bedtime, with a full meal, or on an empty stomach. The compliance implications are significant. Every restriction removed is a barrier to adherence eliminated.
"People living with obesity need treatment options that meet them where they are - and for many, a once-daily pill that can be taken with no food or water restrictions can offer greater flexibility in how they approach their treatment." - Dr. Deborah Horn, Center for Obesity Medicine, UTHealth Houston
The mechanism of action remains the same GLP-1 receptor agonism. Foundayo binds to GLP-1 receptors in the pancreas, gut, and brain. It enhances insulin secretion, slows gastric emptying, and reduces appetite through central nervous system signaling. The difference is in the delivery vehicle, the molecular scaffold, and the practical reality of taking the drug every day for months or years.
The Numbers: What the ATTAIN Trials Actually Show
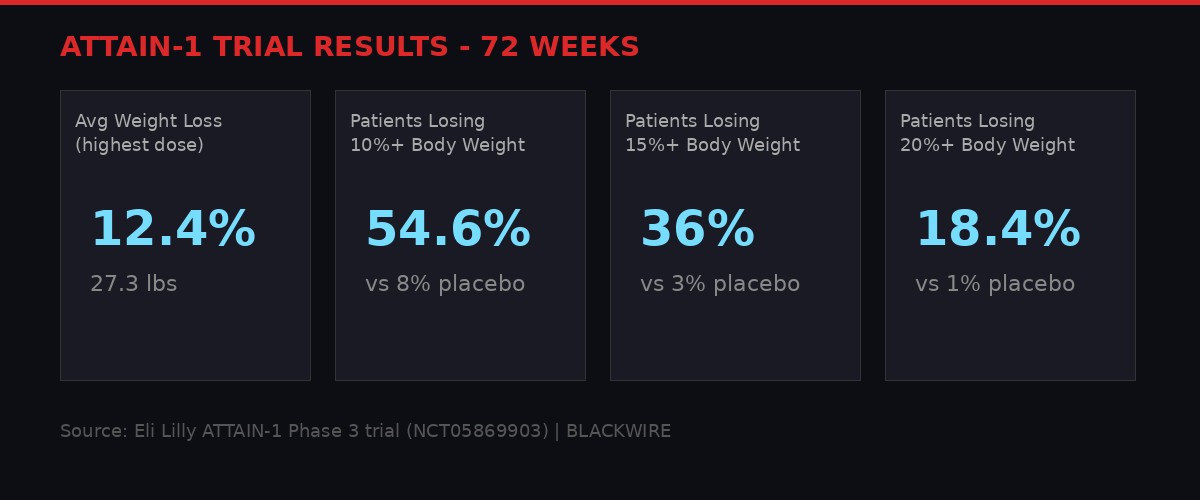
ATTAIN-1 trial results across 72 weeks. Graphic: BLACKWIRE
Foundayo's approval rests on the ATTAIN clinical development program, which enrolled more than 4,500 adults with obesity or overweight across two major Phase 3 trials spanning the United States, Brazil, China, India, Japan, South Korea, Puerto Rico, Slovakia, Spain, and Taiwan.
ATTAIN-1 (NCT05869903) was the pivotal registration trial. It randomized 3,127 participants to receive various doses of orforglipron or placebo over 72 weeks, alongside healthy diet and physical activity counseling. The primary endpoint was body weight reduction from baseline at 72 weeks.
The headline numbers: participants on the highest dose of Foundayo who stayed on treatment lost an average of 27.3 pounds, representing 12.4% of their body weight, compared to 2.2 pounds (0.9%) in the placebo group. In the intention-to-treat analysis, which includes all participants regardless of whether they completed the trial, the highest-dose group lost an average of 25 pounds (11.1%), compared to 5.3 pounds (2.1%) for placebo.
The responder analysis tells a more granular story. Among patients on the highest dose who completed 72 weeks:
- 54.6% achieved 10% or greater body weight reduction - roughly 20 pounds for someone starting at 200 pounds
- 36% achieved 15% or greater reduction - approximately 30 pounds at 200-pound baseline
- 18.4% achieved 20% or greater reduction - exceeding 40 pounds for the same baseline
- Placebo responder rates for the same thresholds were 8%, 3%, and 1% respectively
ATTAIN-2 (NCT05872620) studied orforglipron specifically in adults with obesity or overweight and type 2 diabetes. Over 1,600 participants were randomized across the U.S., Argentina, Australia, Brazil, China, Germany, Greece, India, South Korea, and Puerto Rico. Full results from this trial are expected to support additional regulatory filings globally.
Beyond weight loss, the ATTAIN program documented reductions in waist circumference, non-HDL cholesterol, triglycerides, and systolic blood pressure across all dose groups. These cardiovascular risk markers matter because obesity is not just a cosmetic concern - it is a metabolic disease with systemic consequences.
A separate maintenance study examined what happens when patients switch from injectable GLP-1 drugs to Foundayo. Those switching from Wegovy to Foundayo regained an average of only 2 pounds. Those switching from the more potent Zepbound regained about 11 pounds - expected, given Zepbound's stronger weight-loss profile. The finding suggests Foundayo could serve as a step-down maintenance therapy for patients who achieve their goal weight on injections and want a less intensive long-term option.
The Side Effects: What Patients Need to Know

GLP-1 drugs share a common side-effect profile centered on gastrointestinal symptoms. Photo: Pexels
Foundayo carries the same class-wide side-effect profile that has become familiar to the tens of millions of people now taking GLP-1 medications globally. The most common adverse events reported in clinical trials include nausea, constipation, diarrhea, vomiting, indigestion, abdominal pain, headache, bloating, fatigue, belching, heartburn, flatulence, and hair loss.
The tolerability data from ATTAIN-1 shows a dose-dependent dropout pattern. About 10% of adults on the highest dose discontinued treatment due to side effects, compared to roughly 5% on the lowest dose. These rates are broadly comparable to injectable GLP-1 drugs, though the gastrointestinal side effects tend to be most pronounced during the dose-escalation phase and diminish over time as the body adjusts.
The more serious warnings deserve attention. Like all GLP-1 receptor agonists, Foundayo carries a boxed warning about thyroid C-cell tumors. In rodent studies, GLP-1 drugs have been shown to cause thyroid tumors, including medullary thyroid carcinoma (MTC). It remains unknown whether GLP-1 drugs cause these tumors in humans, but patients with a personal or family history of MTC or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2) should not take Foundayo.
Other serious potential side effects include pancreatitis, severe stomach problems, dehydration leading to kidney complications, gallbladder problems, hypoglycemia when used with insulin or sulfonylureas, and allergic reactions. Patients are advised to watch for symptoms of pancreatitis - severe abdominal pain that doesn't resolve, with or without nausea and vomiting - and contact their healthcare provider immediately.
Hair loss, listed among the common side effects, has become a point of concern in the GLP-1 community. The mechanism is believed to be telogen effluvium - temporary hair shedding triggered by rapid weight loss rather than the drug itself. As the body adjusts to a new weight set point, hair growth typically normalizes, though the experience can be distressing for patients who are unprepared for it.
The Price War: How Foundayo Reshapes the Economics of Obesity Treatment
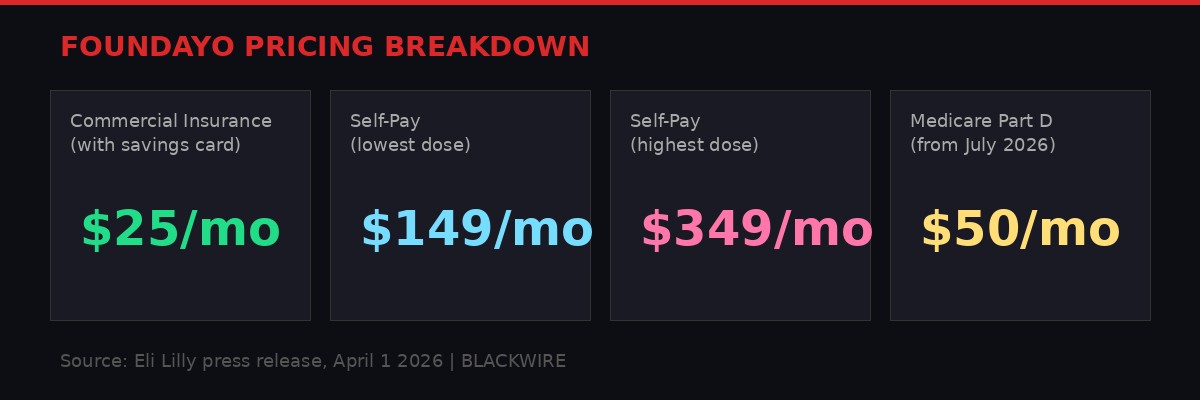
Pricing tiers for Foundayo across insurance categories. Graphic: BLACKWIRE
The economics of GLP-1 drugs have been a flashpoint since Ozempic first went viral on TikTok in 2022. Injectable obesity treatments from both Eli Lilly and Novo Nordisk have carried list prices exceeding $1,000 per month in the United States, placing them out of reach for the vast majority of the 110 million American adults classified as obese by BMI criteria.
Foundayo's pricing structure represents a deliberate attempt to expand the addressable market. The self-pay floor of $149 per month for the lowest dose undercuts the cash price of injectable alternatives by roughly 85%. The $25 per month commercial insurance copay, available through Lilly's savings card program, makes the drug accessible at a price point comparable to many generic medications. And the $50 per month Medicare Part D price, launching July 1, 2026, opens the door to the massive population of seniors with obesity - a demographic that has been largely excluded from GLP-1 therapy due to coverage restrictions.
These price points were not arrived at organically. They are the product of a deal between the Trump administration and both Eli Lilly and Novo Nordisk, struck in November 2025 and formalized through the TrumpRx platform. Under the agreement, both companies committed to specific price ceilings for their obesity medications in exchange for inclusion in the government-backed prescription program and the accelerated FDA review pathway that delivered Foundayo's 50-day approval.
The manufacturing economics of small-molecule drugs further support aggressive pricing. Peptide-based injectables like Zepbound require mammalian cell culture or synthetic peptide chemistry, cold-chain logistics, and prefilled injection devices - all of which add cost. Orforglipron is manufactured using conventional small-molecule synthesis, pressed into standard oral tablets, and shipped at room temperature. Eli Lilly has invested more than $55 billion in manufacturing capacity since 2020, including dedicated facilities for Foundayo production.
"Unfortunately, price is what is driving the decision-making between clinicians and patients for these drugs because they're all excellent drugs and we have lots of options now, but it's still a financial decision at the end of the day." - Dr. Nidhi Kansal, obesity medicine physician, Northwestern Medicine
Analysts at BMO Capital Markets note that Foundayo's lower price point and pill format could attract "casually interested patients" who were unwilling to inject themselves or pay four figures per month. The casual market - people who want to lose 20-30 pounds but don't view their weight as a medical emergency - has been largely untapped by injectable GLP-1 drugs. Foundayo may change that calculus entirely.
For context on the revenue stakes: FactSet analysts project Foundayo will generate $14.79 billion in annual sales by 2030. That sits alongside projections of $24.68 billion for Zepbound and $44.87 billion for Mounjaro in the same year. Combined, Eli Lilly's GLP-1 portfolio could exceed $84 billion in annual revenue by the end of the decade - a figure that would make it the most commercially successful drug franchise in pharmaceutical history.
The Competition: Novo Nordisk, the Wegovy Pill, and the Race for the World

Eli Lilly and Novo Nordisk are locked in the most consequential pharmaceutical rivalry of the century. Photo: Pexels
Foundayo does not enter a vacuum. Novo Nordisk's pill version of Wegovy received FDA approval in December 2025 and launched in U.S. pharmacies in January 2026. It has seen explosive early uptake, with more than 600,000 prescriptions written in March alone and weekly prescription volumes hitting 50,000 within three weeks of launch.
But the Wegovy pill comes with significant practical constraints. Oral semaglutide requires a specific dosing protocol: take it first thing in the morning on an empty stomach with no more than four ounces of water, then wait at least 30 minutes before eating, drinking, or taking other medications. These restrictions exist because semaglutide is a peptide that needs protection from stomach acid and food interference to achieve adequate absorption. The pill uses a permeation enhancer called SNAC (sodium N-[8-(2-hydroxybenzoyl)amino]caprylate) to facilitate absorption through the gastric lining.
Novo plans to argue superiority on efficacy. In clinical trials, the Wegovy pill demonstrated average weight loss of 16.6% at 16 months, compared to Foundayo's 12.4% at 18 months. No head-to-head trial has been conducted, so direct comparison is complicated by differences in trial design, patient populations, and endpoints. But the raw percentages favor Novo's product.
Lilly plans to counter with convenience. No timing restrictions. No food restrictions. No water restrictions. Take it whenever you remember. For a medication that patients will take daily for years, possibly decades, this flexibility matters. Medication adherence is the single largest predictor of treatment failure in chronic disease management, and every dosing hurdle removed incrementally improves the probability that patients will stick with therapy.
Novo Nordisk CEO Mike Doustdar told CNBC in February 2026 that one of the earliest takeaways from the Wegovy pill launch is market expansion. The pill appears to be drawing in new patients rather than converting existing injection users. "The pie is getting bigger, not just being resliced," as one JP Morgan analyst put it. Eli Lilly CEO Dave Ricks agreed, saying Lilly doesn't care whether patients take Foundayo or Zepbound - they want patients on Lilly products, period.
The global dimension is where Foundayo may hold its strongest competitive advantage. Novo's Wegovy pill is currently available only in the United States. Lilly has submitted orforglipron for regulatory approval in more than 40 countries and expects to begin global launches as soon as approvals are granted. Because Foundayo is a small molecule that doesn't require cold-chain distribution, it can be deployed in emerging markets where injection infrastructure and cold storage are limited. The $50 million licensing deal with Chugai in 2018 now looks like one of the most prescient pharmaceutical acquisitions of the decade.
Meanwhile, India has already seen the launch of generic semaglutide injectables at dramatically lower prices. But generic orforglipron is further away - Lilly's patent protection and small-molecule manufacturing processes will take longer to replicate than peptide synthesis. The global market dynamics could favor Foundayo as the first truly scalable oral GLP-1 for worldwide distribution.
The Broader Market: A $150 Billion Industry Reshaping Everything
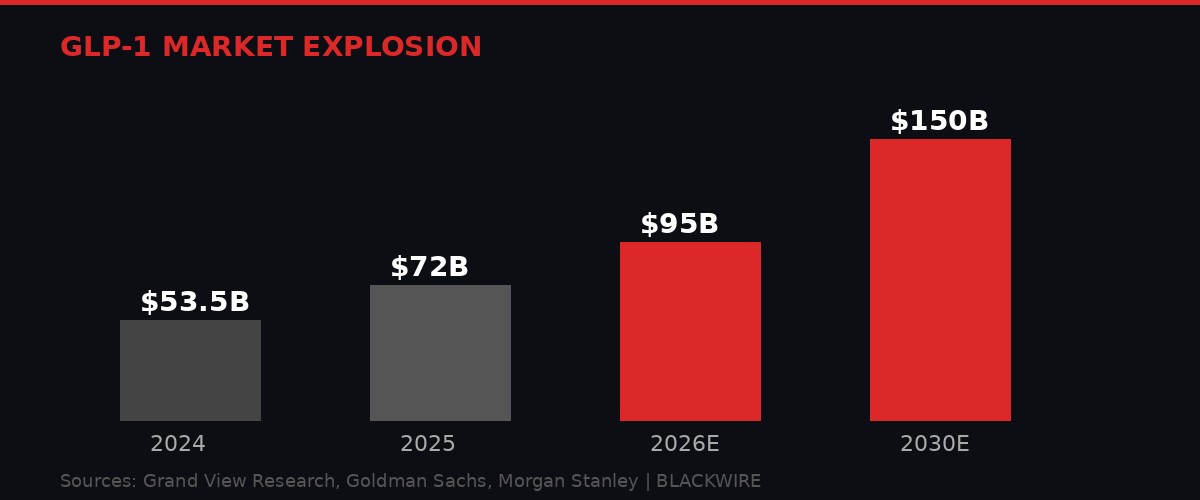
The GLP-1 market is projected to triple by 2030. Graphic: BLACKWIRE
The GLP-1 drug class is not just a pharmaceutical story. It is an economic force reshaping multiple industries simultaneously. The global GLP-1 agonist market was estimated at $53.5 billion in 2024, according to Grand View Research. Goldman Sachs projects the anti-obesity drug market will reach $95 billion by 2030. Morgan Stanley's more aggressive estimate puts the total weight-loss medication market at $150 billion by 2035.
JP Morgan's research division published findings in February 2026 projecting that GLP-1 treatments will lead to an annual revenue reduction of $30 to $55 billion for the food and beverage industry by 2030-2034, as consumers taking these drugs reduce caloric intake by an estimated 21% and grocery spending by 31%. The downstream effects are already visible: Walmart reported in its Q4 2025 earnings call that GLP-1 users are purchasing measurably different grocery baskets - fewer processed snacks, less alcohol, more protein. Fast-food chains have noticed similar patterns.
The insurance and employer benefits landscape is shifting in parallel. As of early 2026, an estimated 43% of large U.S. employers cover GLP-1 medications for obesity, up from roughly 25% in 2024. The Medicare Part D inclusion, beginning this summer, will be the most consequential coverage expansion yet, bringing millions of seniors into the treatment population for the first time.
For Eli Lilly specifically, the Foundayo approval comes at a critical inflection point. The company's stock had fallen approximately 14% year-to-date after a multi-year surge that briefly made Lilly the first healthcare company to reach a $1 trillion market capitalization. Analysts at Cantor Fitzgerald say a successful Foundayo launch is key to stock recovery. They'll be tracking weekly prescription volumes as the leading indicator.
"It's a big moment. We've been working in this category of medicines for 20 years. Here is an option that's more accessible, easier to fit into your daily routine." - Dave Ricks, CEO, Eli Lilly
Lilly has another card to play. Retatrutide, its next-generation injectable obesity drug that targets GLP-1, GIP, and glucagon receptors simultaneously, is in late-stage clinical trials for weight loss. Early data has shown weight loss exceeding 24% - approaching bariatric surgery levels without the operating room. If retatrutide delivers on its promise, Lilly will have assembled a portfolio spanning entry-level oral therapy (Foundayo), mid-tier injectable (Zepbound/Mounjaro), and a premium next-generation shot (retatrutide) - covering every segment of the obesity treatment market.
The Bigger Picture: Obesity as a Chronic Disease, Finally

Foundayo's accessibility may finally bring obesity treatment into mainstream primary care. Photo: Pexels
For decades, obesity has been treated as a moral failing rather than a medical condition. Eat less, exercise more, try harder. The arrival of effective pharmacotherapy - first the injections, now the pills - is forcing a reckoning with that narrative. Fewer than one in ten people who could benefit from a GLP-1 medication are currently taking one, according to Eli Lilly's own estimates. The barriers are not just cost and access. They include stigma, provider reluctance, and the deeply embedded cultural belief that weight is a matter of willpower.
The injection format itself has been a psychological barrier. As Lilly executive Ken Custer explained: "Beyond supply and affordability, one of the bigger barriers to adoption has been that some patients just don't want to take an injection. That could be because it's a needle, but it also may just be that for them, an injection signifies that their condition is more severe than they feel it is at that point."
A pill changes that calculus. Pills are familiar. People take pills for blood pressure, for cholesterol, for allergies. There is no stigma associated with swallowing a tablet at breakfast. For the millions of Americans who are 30-50 pounds over a healthy weight but would never consider injecting themselves with a medication, Foundayo may represent the tipping point.
The Obesity Action Coalition, a patient advocacy organization, greeted the approval cautiously but optimistically. "There is no single path that works for everyone living with overweight or obesity," said Joe Nadglowski, the group's president and CEO. "New treatment options expand choice and help more people find care that fits their lives, their goals, and where they are in their journey."
The implications for the U.S. healthcare system are enormous. Obesity is linked to type 2 diabetes, cardiovascular disease, certain cancers, sleep apnea, joint disease, and depression. The CDC estimates that obesity-related healthcare costs in the United States exceed $173 billion annually. If even a fraction of the eligible population begins treating obesity with GLP-1 medications, the downstream reduction in comorbidity-related healthcare spending could be substantial - though the timeline for those savings to materialize remains uncertain.
Foundayo is also being studied for type 2 diabetes, obstructive sleep apnea, osteoarthritis knee pain, hypertension, peripheral artery disease, and stress urinary incontinence. The drug's potential to treat multiple conditions through a single daily pill could simplify treatment regimens for patients managing several obesity-related comorbidities simultaneously.
The FDA Fast Track: 50 Days and What It Means

The FDA's 50-day review was the fastest obesity drug approval in history. Photo: Pexels
The speed of Foundayo's approval is itself a story. Standard FDA drug approvals take six to ten months. Priority reviews take roughly six months. Foundayo was reviewed in 50 days under a new pilot program called the Commissioner's National Priority Voucher (CNPV) program, designed to expedite approvals for drugs that address national health priorities.
Obesity qualifies. More than 42% of American adults are classified as obese by BMI. The condition is the second-leading cause of preventable death in the United States after smoking. The economic burden on the healthcare system is staggering. And the current treatment landscape - dominated by expensive injections with constrained supply - leaves the vast majority of eligible patients untreated.
The 50-day timeline does not mean the FDA cut corners. Eli Lilly submitted a complete New Drug Application with full Phase 3 trial data from the ATTAIN program. The FDA reviewed the same volume of clinical evidence it would review under standard timelines. The acceleration came from prioritized reviewer assignments, parallel review tracks, and pre-submission meetings that resolved potential questions before formal filing.
Critics have raised concerns about the optics of fast-tracking a drug from a company that struck a pricing deal with the Trump administration. The CNPV program was created under the current FDA commissioner, and its first use on a drug from one of the two companies in the TrumpRx agreement raises questions about the relationship between commercial negotiations and regulatory process. The FDA has stated that the review was conducted independently of any pricing agreements and that the clinical data supported approval on its merits.
Regardless of the political dynamics, the precedent is set. If a 50-day approval is possible for obesity drugs, other therapeutic areas - Alzheimer's disease, antibiotic resistance, rare cancers - may push for similar treatment. The CNPV program could reshape the pace of pharmaceutical innovation in the United States if it survives administrative transitions and legal challenges.
Timeline: From Chugai Lab to American Medicine Cabinets

The eight-year journey from licensing deal to FDA approval. Photo: Pexels
- 2018: Eli Lilly licenses orforglipron from Chugai Pharmaceutical (Japan) for $50 million upfront
- 2022: Phase 1 trials demonstrate proof of concept for oral GLP-1 activity
- 2023: Phase 2 trial results published - 36-week data shows up to 14.7% body weight reduction with no apparent plateau
- June 2023: ATTAIN Phase 3 program begins enrollment - 4,500+ participants across 10+ countries
- November 2025: Eli Lilly and Novo Nordisk strike pricing deal with Trump administration via TrumpRx platform
- December 2025: Novo Nordisk's Wegovy pill receives FDA approval - first oral GLP-1 for obesity
- January 2026: Wegovy pill launches in U.S. pharmacies - hits 50,000 weekly prescriptions within 3 weeks
- Early February 2026: Eli Lilly submits Foundayo NDA under Commissioner's National Priority Voucher program
- February 2026: Eli Lilly reports Q4 earnings - Zepbound and Mounjaro revenues surge, stock under pressure from broader market
- March 2026: Novo reports 600,000+ Wegovy pill prescriptions in a single month
- April 1, 2026: FDA approves Foundayo - 50-day review, fastest obesity drug approval ever
- April 6, 2026: Foundayo begins shipping via LillyDirect with free home delivery
- July 1, 2026 (expected): Medicare Part D coverage at $50/month launches
- 2026-2027: Regulatory filings in 40+ countries - global rollout begins
What Happens Next

The GLP-1 pill market is just getting started. Photo: Pexels
The immediate question is uptake velocity. Novo Nordisk's Wegovy pill hit 600,000 prescriptions in March. If Foundayo can match that pace within its first full month of availability, the combined oral GLP-1 market will be outpacing most analysts' projections before summer. The no-restriction dosing protocol could give Foundayo an edge in patient satisfaction and adherence, but Novo's three-month head start and stronger efficacy data provide a meaningful counterweight.
Insurance coverage decisions over the next 90 days will be critical. Pharmacy benefit managers and health plans that already cover injectable GLP-1 drugs will face pressure to add Foundayo to their formularies, especially given its lower price point. Employers who have resisted covering $1,000-per-month injections may find $149-per-month pills harder to exclude. Every formulary addition expands the addressable market.
The global rollout will test whether Foundayo can deliver on its scalability promise. Lilly has filed in 40+ countries and plans to launch as soon as each approval is granted. For developing nations where injectable drug infrastructure is limited - much of sub-Saharan Africa, Southeast Asia, parts of Latin America - a room-temperature oral GLP-1 could be transformative. Global obesity rates have tripled since 1975. The need is not confined to wealthy nations.
And then there's retatrutide. If Lilly's triple-agonist injectable delivers Phase 3 data consistent with its earlier readouts - 24% or greater weight loss - the company will control the entire spectrum of obesity pharmacotherapy. Entry-level pill. Mid-range injection. Premium triple-agonist. The strategy mirrors what Apple did with the iPhone lineup: a product at every price point, all feeding the same ecosystem.
The needle isn't dead yet. Injectables will remain the gold standard for maximum weight loss for years to come. But the needle's monopoly on GLP-1 therapy ended on April 1, 2026. Foundayo is the first shot - or rather, the first pill - in what will be a decade-long transformation of how the world treats its most prevalent chronic disease.
For the 800 million adults worldwide living with obesity, according to the World Health Organization's 2025 estimates, the question is no longer whether effective treatment exists. It's whether they can get to it. A pill that costs $149, ships to your door, requires no injection training, no refrigerator space, and no empty-stomach alarm clock at 6 AM represents the closest thing to an answer the pharmaceutical industry has ever produced.
Foundayo prescriptions are open now. Shipping starts April 6. The obesity drug war just entered its oral phase.
Get BLACKWIRE reports first.
Breaking news, investigations, and analysis - straight to your phone.
Join @blackwirenews on Telegram